As many of you will know, we are running the DevelopAKUre clinical trials. These should give us evidence whether nitisinone is effective as a treatment for AKU. This weeks blog explains the third part of our clinical trial, SOFIA.
DevelopAKUre is made up of three parts:
- SONIA 1: this first study looked at different doses of nitisinone to determine which has the best effect. This study concluded in 2013 and set the dose of nitisinone at 10mg per patient per day. (However, the dose given at the NAC will stay the same until DevelopAKUre has finished.)
- SONIA 2: the second study is ongoing, and due to finish in 2019. We have successfully recruited almost 140 patients across Europe. Half of those patients receive nitisinone, and half do not. The study aims to compare the two groups after four years and assess the effects of nitisinone.
- SOFIA: this is the third part of DevelopAKUre and our focus for 2016.
What is SOFIA?
SOFIA stands for “Subclinical Ochronotic Features in Alkaptonuria”. Ochronosis is the process where cartilage is discoloured black by the effects of AKU. It’s thought to be the earliest sign of the more damaging aspects of AKU. Therefore, SOFIA will study different ages of patients to see at what age ochronosis starts, and therefore better predict the time before joint damage begins. This is vital to know, so that we can best decide at which age to begin treatment with nitisinone.
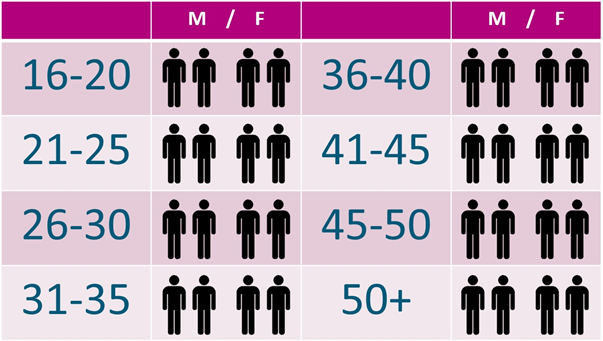
We aim to recruit 32 patients in SOFIA, from a range of ages. The chart below shows our aim to find 2 male and 2 female patients for each 5-year age range from 16 to 50+.
So far 17 people have already successfully completed the SOFIA study.
What will SOFIA involve?
Patients enrolled into SOFIA will need to visit the clinical site in Liverpool, UK, for one single visit. The visit will last three days, and all travel and accommodation expenses for patients will be covered by the study. A translator will be available if you don’t speak English.
During the visit, several tests will be performed, including: urine tests, blood tests, photos of eyes and ears, questionnaires, an MRI scan, ear biopsy and gait analysis. The ear biopsy involves taking a small piece of cartilage from the back of the ear after a local anaesthetic injection. This should be relatively painless, and will not leave a scar. This cartilage will then be examined to look for early signs of discolouration. The gait analysis measures how patients’ walk, by using special cameras to record their speed and position of joints. This will show if the patient is starting to experience joint pain, as even small changes to the joint will change how they walk.
How does the AKU Society help?
Once an AKU patient has agreed to take part in SOFIA, the AKU Society will liaise with the hospital and the patient to pick preferential dates for the visit to Liverpool. We will aim to fit travel to Liverpool with the patient’s personal and work commitments. Once we have a date, the AKU Society will then contact Liverpool and ensure all flights, hotel rooms and taxis are booked.
We will be available to support patients in every aspect of their participation in SOFIA and can assist patients with communicating to the hospital and will liaise with Liverpool to make your participation in SOFIA as easy as possible.
Where to get more information?
The AKU Society will contact eligible patients to discuss enrolment into SOFIA. If you are an AKU patient who is keen to take part, you must not be taking nitisinone and must be aged more than 16 years old. We’re looking for patients who live in Europe, and are able to travel to the UK. Please contact ciaran@akusociety.org for more information.
