Alkaptonuria (AKU) was the first condition noted as following Mendelian Inheritance by Sir Archibald Garrod in 1902. ¹
Since then, more work has been done to understand its genetic mechanism. AKU is now understood to be a recessive disorder, caused by a single gene defect. It is mapped to Chromosome 3 between regions 3q21-q23, ² the site of the homogentisate 1,2-dioxygenase (HGD) gene.³
More than 80 mutations ⁴ in the HGD gene have been identified in people with the disease. Many of these mutations lead to changes in single amino acids in the homogentisate dioxygenase protein. Mutations in the HGD gene probably deactivate the enzyme by changing its structure.
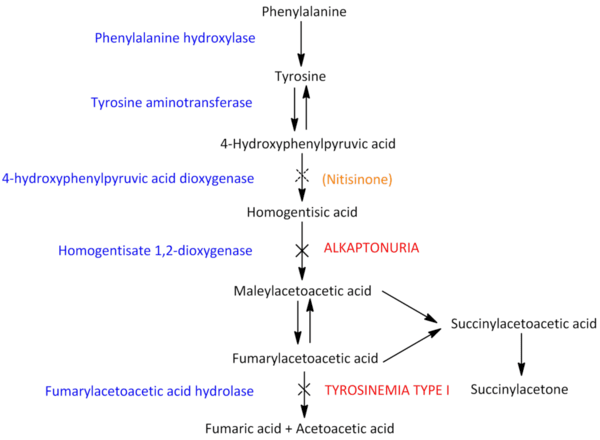
HGD is a vital enzyme in tyrosine metabolism. The graphic below shows a general overview of the pathway. With a malfunctioning or inactive HGD enzyme, alkaptonuria patients are unable to convert homogentisic acid (HGA) ⁵ into maleylacetoacetic acid. ⁶ Instead of the natural condition, in which excess tyrosine is eliminated from the body, AKU patients end up converting excess tyrosine into HGA. ⁷
In the body of an AKU patient, HGA accumulates at more than 2,000 times the normal rate.⁸ Some of this is excreted in the urine, but a significant quantity remains within the body. HGA turns black on oxidation, leading to black urine and occasional black sweat which can be indicative of alkaptonuria. A small proportion of patients, however, will not develop these symptoms.
Much of the research into AKU has focused on the urine. It was discovered that the darkening was delayed in an acidic solution and accelerated in alkaline conditions. This affinity for alkali led Boedeker, in 1859, to describe the urine as containing an alkapton, later leading to the name alkaptonuria.
Through a simple test with Benedict’s sugar reagent, it was noted that the urine was a powerful reducing agent. This not only reduces the copper reagent to an orange precipitate, but also darkens the solution due to its alkalinity. The net effect is orange particles suspended in a muddy brown
solution.
If you would like any further information, or have a patient with AKU, please contact hannah@akusociety.org
E-learning modules for healthcare professionals
We have developed online eLearning modules all about AKU with the RCN and RCGP. The aim of these modules is to increase awareness of AKU among health professionals, especially GPs, and ultimately to improve diagnosis.

Royal College of General Practitioners
This course will deliver an understanding of alkaptonuria (AKU). It will introduce GPs to this rare condition and aim to improve their knowledge of the presentation and management in primary and secondary care, as well as referral pathways to the national centre in Liverpool.

Royal College of Nursing
This learning module focuses on alkaptonuria (AKU) or black bone disease, a rare genetic disease that prevents the body from breaking down the amino acids, tyrosine and phenylalanine. It introduces nurses to the condition and explains how to recognise it, which can lead to earlier diagnosis and allow patients access to timely disease-modifying therapy.
